So, I gushed a little bit about this new long-acting injectable (LAI) version of risperidone called Uzedy in Impressions from the 2024 American Psychiatric Conference. From a practical perspective I really do think there are things to be excited about here. Most LAIs on the market are a bit cumbersome to use in one way or another. All of them involve injections of thick, viscous liquids1 into a muscle which means long, large needles. Some — like Invega and Aristada — require multiple closely-spaced injections in the beginning of treatment. Haloperidol is a quagmire of various different dosing schedules and oral supplementation timelines. Zyprexa Relprevv is barely ever used because of the 3 hour post-injection monitoring period.
A LAI that is effective after a single injection, with a small injection volume given subcutaneously (read: doesn’t need a big needle), that can be given every 1-2 months is a meaningful improvement. I think that part of my role as a physician is to give props to pharma companies when they make cool, clinically useful things instead of more useless copycat drugs.
That said, I didn’t take much time to look at their efficacy data in any sort of detail. I feel badly about this because it is also my role as a physician to make sure that I don’t let the pharmaceutical companies’ cool biopolymers distract me from what actually matters: Making it to the top of CMS’s OpenPayments leaderboard for psychiatry by unseating Stephen Stahl!
Er, sorry, I meant to write “optimal patient care,” but my fingers slipped.
I’m going to walk through their Phase 3 trial of the drug formerly known as TV-460002: Efficacy and safety of TV-46000, a long-acting, subcutaneous, injectable formulation of risperidone, for schizophrenia: a randomized clinical trial in the USA and Bulgaria
Uzedy? I’m zedy3.
Study Design
Endpoints
Perhaps the most important place to start, because they frame the entire study. Primary and secondary endpoints were pre-registered4 (i.e. set prior to seeing the outcome data), which is standard practice for clinical trials nowadays, but always good to note.
The primary endpoint was time to impending relapse in the intent-to-treat population. ‘Impending relapse’ is confusingly defined as meeting one of the following criteria:
Criterion A:
CGI-I ≥ 55
-AND-Select PANSS Score ≥ 5 for 1 of 4 specific items6 with:
+2 points from randomization on any one item
-OR-
+4 points summed across all four items
Criterion B: Hospitalization due to worsening psychosis
Criterion C: CGI-SS7 Part 1 score ≥ 4 -OR- Part 2 score ≥ 6
Criterion D: Violent behavior
The authors say that this is a common criteria in LAI studies and cite this paper, but I can’t even find the phrase “time to impending relapse” in it. I went and looked at papers for other LAIs and they all use pretty similar criteria. The Uzedy criteria seem to be more conservative than average, which is a point in its favor.
I am curious about why they singled out these four PANSS items: conceptual disorganization, hallucinatory behavior, suspiciousness, unusual thought content. Using some subset of PANSS items doesn’t seem to be unusual though, and other LAI studies I looked at used their own proprietary blends of PANSS items. I talked this over with another psychiatrist friend and we think the most likely reason these four were selected is because they are:
Positive symptoms8 (i.e. the target of antipsychotics)
Relatively specific to schizophrenia (the remaining positive items are excitement, hostility, and grandiosity)
Relatively easy to observe
Recruitment and Demographics
Patients were recruited across 69 clinical sites across the USA and… Bulgaria for some reason? No idea why Bulgaria. Can’t find anything that explains why, and neither the paper nor the supplement do any sort of analysis based on country.
Subjects were required to be between 18-65 in the Bulgarian cohort or 13-65 in the American cohort, but the Americans only managed to recruit a single 17 year-old so this ends up being irrelevant9.
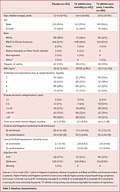
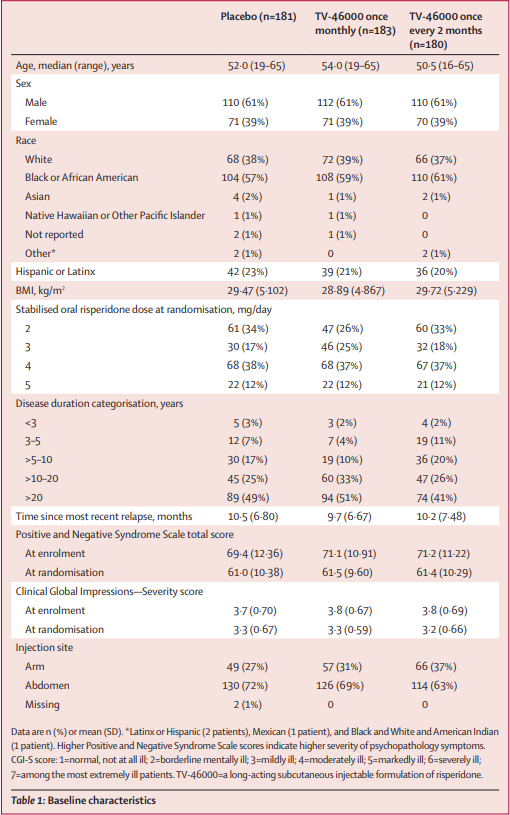
I think it can be helpful to look at demographics tables and summarize the study population to help avoid saying overly broad things like “this is a study done in which the subjects were schizophrenics.” In this case, this is primarily a study of:
Chronically ill, non-treatment resistant schizophrenics
With mild/moderate illness
Whose illness is known to respond to risperidone
Major Inclusion Criteria
Had a diagnosis of schizophrenia for least 1 year prior to screening (98% of study patients carried the diagnosis for at least 3 years)
At least one relapse within the last 2 years.
‘Response’10 to an antipsychotic agent other than clozapine within 1 year of screening
PANSS score <100 at screening
I just want to stop here for a second and point out the enrichment going on here. First, the study is only picking individuals who have had a response to antipsychotics in the past. Strictly speaking, this study does not answer whether or not Uzedy is effective in previously untreated individuals, though I would be hard pressed to come up with a reason why it wouldn’t behave similarly to oral risperidone/paliperidone in those individuals.
Second, the PANSS ceiling of 100 means that really sick patients were not eligible for inclusion. At enrollment, the median PANSS score was ~70 with a std. dev. of ~11 points. With 863 individuals at time of enrollment, that means that only about 39 (~4.5%) of these patients would’ve had scores between 92-100.
Certainly this is the authors stacking the deck in their favor, but in this case the game is already rigged against them in 50 different ways, so I think it’s reasonable that we let them cheat at least a little bit. Very ill schizophrenics are not the sort of people who are willing to let a doctor inject them with an experimental drug and then dutifully follow up for surveillance and additional injections over the next 52 weeks.
It’s worth remembering that the average dropout rate in clinical trials — of people who mostly do not believe that shady government entities are trying to implant microchips in them — is somewhere around 20%. This study started out with 863 enrolled subjects and only 339 (39%!) remained in the final analysis.
Finally — like I pointed out above — for this critique to really have teeth, you’d have to make a good argument as to why Uzedy would be uniquely worse than oral risperidone/paliperidone in individuals with higher PANSS scores. Three paragraphs later and I’ve still got nothing.
Major Exclusion Criteria
Clinically significant DSM-5 diagnosis other than schizophrenia
Current treatment with clozapine
ECT within 1 year before screening
Risk of violent behavior per clinical judgement
Risk of suicide per clinical judgement or C-SSRS ≥ 3
Nothing all that interesting here. The clozapine and ECT criteria, I think, are just an easy way to exclude “treatment resistant” schizophrenics.
Assuming they got through these filters, now we move on to:
Stage 1
Participants were started on oral risperidone between 2-5mg qDay11 — dose determined by their prescriber — for 12 weeks. If they were on a different antipsychotic they were switched to risperidone. This was done for everyone, regardless of the group they would be randomized to. They were seen in clinic every 4 weeks and got a phone call weekly.
Participants could only progress to stage 2 if they “adhered to study protocol,” took at least 80% of their oral risperidone doses, and were stable for at least 4 consecutive weeks before the baseline randomization visit. “Stability” was defined as:
Outpatient status
PANSS ≤ 80
Score of ≤ 4 on the four select PANSS items listed above12
CGI-S ≤ 4 (moderately ill)
CGI-SS ≤ 2 (mildly suicidal) on Part 1, and ≤ 5 (minimally worsened) on Part 2
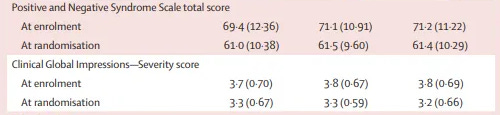
Of the 863 enrolled, only 514 progressed to Stage 2. Again, we’re enriching the study by cutting out the patients that are not responsive to risperidone. I think this probably best explains the drop in PANSS and CGI-I scores at randomization that we see in the demographics table13.
So, participants who take their risperidone and are stable for ≥4 weeks prior to week 12 of Stage 1 then move on to…
Stage 2
Participants were randomized 1:1:1 by sex and oral risperidone dose to receive Uzedy injections monthly, every two months, or placebo injections. The dose of Uzedy received was equivalent to the dose of risperidone they were stabilized on in Stage 1. Patients in the q2Month group still got injections every month, but one of them was a placebo injection so they wouldn’t know they were in the q2Month group.
Dose adjustments were not permitted, which I think keeps the analysis clean.
The biggest issue with the transition to Stage 2 is that Teva claims that you do not need oral loading doses with Uzedy. These subjects, however, went straight from oral to the injectable without a washout period, which means that this study can’t back up their claims that there is no clinical need for PO supplementation.
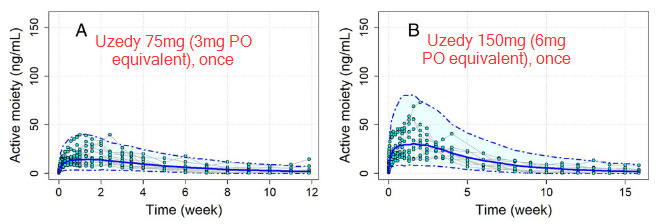
Now, to be fair to Teva, they wrote a paper using data from their Phase 1 study where they measured actual plasma levels in 97 individuals with schizophrenia or schizoaffective disorder and did some simulations. The math mostly seems to check out. Unfortunately, that paper doesn’t have graphs that properly show plasma levels very early after administration, but these simulations of D2 receptor occupancy — which are based on their measured plasma levels and known D2 occupancy levels from other studies — do a good enough job of convincing me that you’re getting to therapeutic levels very fast in most patients.
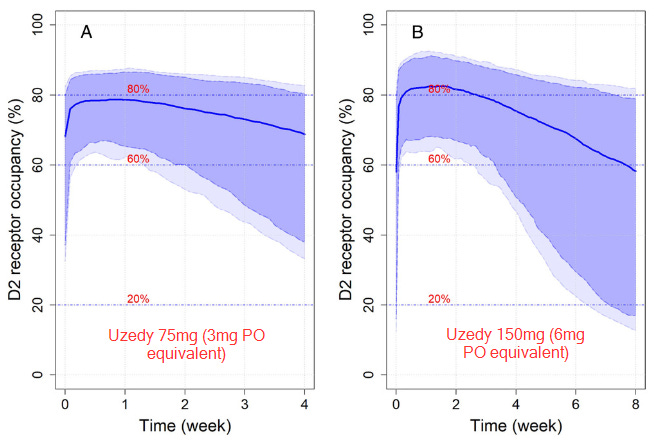
Honestly, I don’t think this is that big of a deal in the grand scheme of things. In most situations you will probably be transitioning from some PO risperidone dose directly to Uzedy anyway. In the event that you aren’t and are nervous, you could just do what you’d do with the aripiprazole LAI and give a single PO dose on the day of injection.
Results
Overall, 62% (339) of subjects completed the study and dropout rates were pretty even between treatment arms.
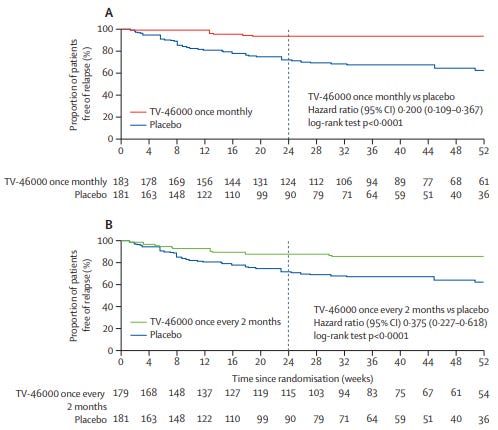
The primary outcome results for Uzedy were actually quite good! In the qMonth group, time to impending relapse was 5 times longer (HR 0.2, 95% CI 0.109-0.367, p<0.0001), and risk of relapse was decreased by 80%. In the q2month group, time to relapse was 2.7 times longer (HR 0.375, 95%CI 0.227-0.618, p<0.0001), and risk of relapse was reduced by 62.5%.
The survival curves separate out nicely as you might expect:
That said, I am totally confused by the survival numbers below each of these graphs. They are clearly the number of individuals who are relapse free at any given timepoint (the starting values are the n’s of the groups as stated in the paper), but the sum of the final numbers at 52 weeks is 15114, less than half of the 339 the paper says “completed” the study. Even if I assume that the “completion” number includes patients who ended early due to impending relapse and treatment discontinuation due to adverse events, that still only gets me to 286. Help!
They used a 30-item 4-point symptom scale called the “Schizophrenia Quality of Life Scale” which asks questions about psychosocial factors, energy/motivation, and symptoms/side-effects to get a sense of whether or not the patients themselves noticed an improvement in their lives overall; the maximum score is 120, lower scores are better. Annoyingly, the authors don’t give the baseline scores for these patients, they just report the total change from baseline to the endpoint. Of the patients who stayed to the end of the trial, the qMonth group had a -5.4 point change, the q2month -4.5 points, and placebo had a 1.14 point increase.
There are pretty small changes in perceived qualify of life, despite what I would consider a very clinically meaningful reduction in relapse rates.
There weren’t any adverse events that are worth spending time on; other than injection site nodules, they were all things that you would expect from giving someone an antipsychotic. There were 5 deaths total, none of which were considered to be medication related.
Overall, the results seem to speak for themselves. Uzedy is unambiguously more effective than placebo in preventing relapse in schizophrenia and seems to be just as efficacious as oral risperidone.
But.
Why does the qMonth look better than q2Months?
This is worth thinking about a little bit for situations where both options seem equally good, and I think is a good illustration of how first principles can sometimes be useful to answer these questions.
Might this be a problem with the pharmacokinetics? Maybe 8 weeks is just too long to wait between doses? Let’s try and think this through, again with the help of the Phase 1 data and Teva’s modeling.
The dots in this figure are actual measurements from the phase 1 trial, the solid blue line is the predicted median curve, and the dashed lines indicate the 5th and 95th %tile prediction boundaries.
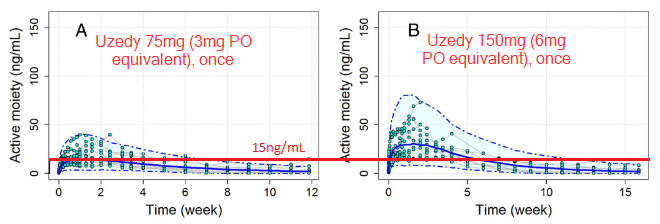
The first question I think is useful to ask is to what degree are individuals in each treatment arm falling below the therapeutic threshold of 15ng/mL, based on these guidelines from the American Society of Clinical Psychopharmacology.15
If you look at plot B, past week 5-6 it sure looks like almost everyone is under the therapeutic threshold, while the qMonth group is close to 50/50 at 4 weeks when they would be due for their next injection.
This could explain what’s going on with the q2month group. Early on in dosing q2months may be just infrequent enough that they’re decompensating. Eyeballing the survival curves, you can see that a big chunk of the relapses for the q2month group are happening before week 8, which is what this hypothesis would predict. There’s also the fact that the longer the study goes on, the fewer relapses we see.
Normally, you could make the argument that this is just because there’s a sort of selection-bias causing this — only people who respond well to risperidone are left standing past the first half of the study. That argument isn’t nearly as strong here, though, because the study ensured that they were stable on oral risperidone for at least 4 weeks prior (which is plenty of time to see a major effect).
Is this just a dose problem?
Maybe the q2Month relapses were concentrated in the low-dose cohorts? Unfortunately, neither the paper nor its supplement provides data stratified by dose.
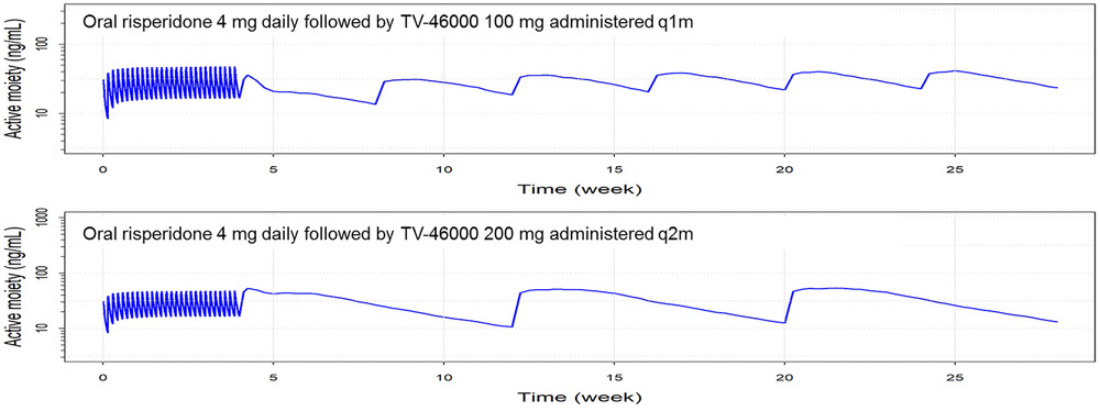
Maddeningly, Teva’s phase 1 trial only collected hard data for qMonth dosing and simulated everything else. However, even their simulations suggest that there might be an issue with the q2month formulation, particularly in patients who clear risperidone more quickly than average. Take a look at these simulated graphs for the 100mg and 200mg formulations (4mg PO equivalent):
Important to note that the blue line here is their median prediction; the caption for this graph says that there should be a light-blue area that indicates a 90% CI, but looks like that just never got included? Also the scaling is different on the y-axis in both. In any case, you’ll note that just before the q2month injection, the median prediction is just about touching the 10ng/mL line. That suggests that fully half of patients are below even their questionable 10ng/mL therapeutic threshold at 8 weeks post-injection! I suspect that if this graph showed the 90% CI, we would see that there is a large proportion of patients who would be under the more clearly supported 15ng/mL threshold even earlier in the curve.
The qMonth injection, on the other hand, doesn’t get nearly as close (despite the difference in scaling), and more quickly moves towards its steady state with repeated injections.
Putting a Bow on It
Obviously we’re going to need some better data to determine whether or not this difference between qMonth and q2Month formulations are just statistical artifact or real differences. In the meantime though, I would look askance at decisions to use the q2Month dosing — initially — unless you have a good reason.
That said, if you’re thoughtful I don’t see why patients couldn’t transition to q2month eventually by using the qMonth dosing for a couple of months (2-3?) to get their trough levels comfortably above 15ng/mL, then switch to the q2Month. This is basically what the paliperidone LAI (Invega) does with its progression from qMonth → q3Month → q6Month formulations.
If you really wanted to start with the q2Month, you could probably get creative with oral supplementation, but that seems like a lot of unnecessary guesswork.
Most formulations conjugate the drug to a long-chain fatty acid (e.g. palmitic acid, decanoic acid, lauric acid) that forms a depot which slowly releases the drug molecule over time.
It’s at least a little bit funny that the research name of this drug implies that the participants are being injected with 46,000 TVs, exactly the sort of absolutely untethered delusional content you’d expect to hear from a schizophrenic
I’m also sorry about that one. It’s an awful brand name for a drug.
It’s important to know that if your primary outcomes don’t reach statistical significance, that’s the end of your analysis; at that point you’re not allowed to talk about significance for any of your secondary endpoints. This might seem like a weird, artificial constraint. It is, but it’s there to prevent researchers from p-hacking by coming up with dozens of different endpoints and cherry-picking significant p-values.
i.e. “Minimally worsened” or worse
These were: conceptual disorganization, hallucinatory behavior, suspiciousness, unusual thought content.
Why these four? I don’t know. I’m guessing that they are the items in the PANSS that are most responsive to antipsychotics.
Clinical Global Impression - Severity of Suicidality
Part I asks what the most severe suicidality was in the last 7 days. 4 = “severely suicidal”
Part II asks how different their suicidality is from baseline. 6 = “much worse”
Yeah I know “unusual thought content” is technically a “general psychopathology item” but it’s a positive symptom in my heart.
Not terribly surprising though. Prevalence of schizophrenia in people under <18 is pretty small, maybe 0.25%?
Response was defined by ‘investigator judgement”
For those of you not familiar with the medical lingo, every time you see a ‘q’ in front of something, it denotes frequency, from the Latin quaque meaning ‘every.’ Thus qDay = daily, qMonth = monthly, etc.
Again, those were: conceptual disorganization, hallucinatory behavior, suspiciousness, unusual thought content
I was also wondering if there was somehow a significant proportion of patients who were untreated prior to enrollment, but seemed unlikely.
61+54+36
Teva claims that the threshold is actually 10ng/mL, but their citation for this is “Data on file. Parsippany, NJ: Teva Neuroscience, Inc.”, so forgive me for not taking them at their word


Wait how can you say this "seems to be just as efficacious as oral risperidone"?
I don't see how this trial answers that question.
Great write up, thank you