Same As It Ever Was
No Surprises In The 52-Week Open-label Cobenfy Study
We have new Cobenfy (Xanomeline/Trospium) data, so I have a new analysis.
I’ve been saying that Xanomeline is a very average looking antipsychotic in terms of efficacy, but is incredibly attractive when it comes to side-effects: No EPS, no weight gain, no metabolic derangements! My feeling has been that if this continues to be observed in longer-term studies, Xanomeline will probably be an excellent first-line treatment for mild/moderate cases of psychosis (i.e. the ones we use aripiprazole for now). Fortunately for me, EMERGENT-5 is chock-full of “safety data” (i.e. side-effect profiles), so let’s get to it.
Methods
This was a Phase 3, multicenter, open-label trial (NCT04820309) conducted at outpatient sites. The study period was 1 year. Of 1,065 individuals screened, 566 completed screening, received at least one dose of xanomeline, and were included in the safety population. 558 of those 566 had a baseline and at least one postbaseline PANSS assessment and were included in the mITT population for efficacy analyses.
Inclusion Criteria
Full inclusion/exclusion criteria are available at ClinicalTrials.gov
Adults with a confirmed diagnosis of schizophrenia
Psychiatrically stable symptoms (PANSS total score ≤80 and CGI-S score ≤4)
No prior exposure to xanomeline/trospium
Currently on antipsychotic medication at the time of enrollment
The patient in EMERGENT-5 is a stable outpatient; someone who is already doing reasonably well on their current antidopaminergic and is being asked to switch to a medication that is completely new to them.1 The mean baseline PANSS total score was 66 (σ: 10.4) and the mean CGI-S was 3.4, putting the average participant somewhere between “mildly” and “moderately” ill. This is a very different population from the EMERGENT 1-3 trials which enrolled acutely psychotic, hospitalized patients with PANSS scores between 80-120.
It is also worth noting that this study does not have a placebo arm. There is no comparator of any kind, which makes it considerably harder to draw conclusions about efficacy. Any improvement in PANSS scores could reflect genuine drug effect, regression to the mean, natural course of illness, or the (presumably considerable) placebo effects we already know exist in this population.
Interventions
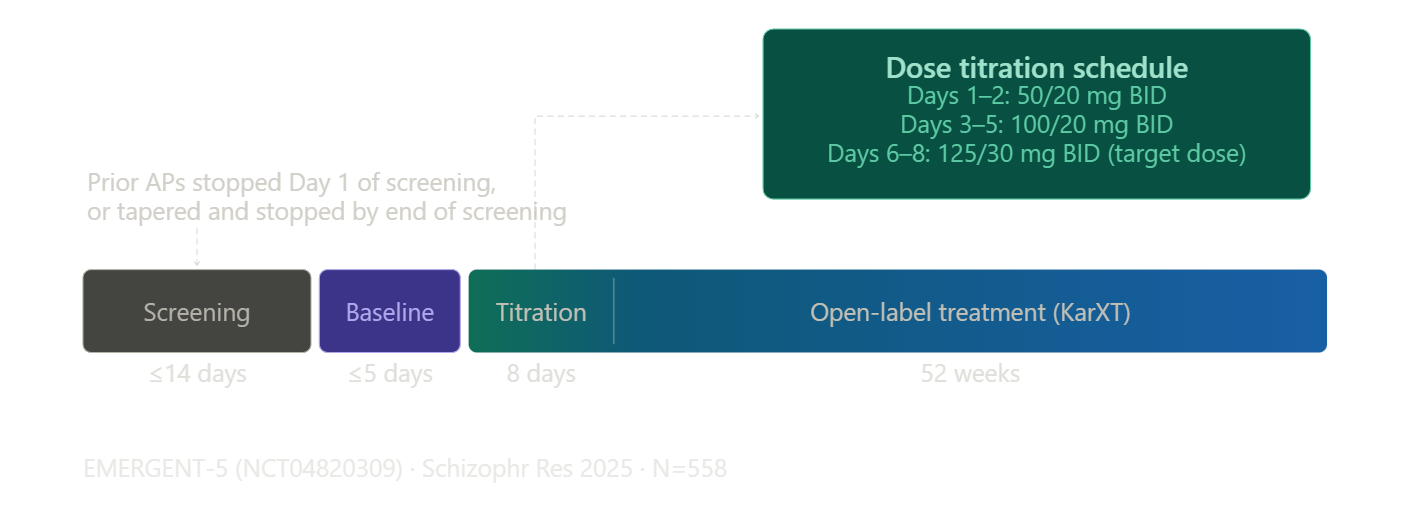
Here’s an overview of the timeline:
Prior antipsychotics were discontinued before starting Xanomeline. They were allowed a down-taper during the 14-day screening period and had to stop their prior antidopaminergic completely before entering the 5-day baseline period. This is long enough for a total washout of most antidopaminergics, except for the partial-D2 agonists.
Anyone on a LAI needed to have had their last injection more than 12 weeks prior, or more than 24 weeks if they were on Invega Trinza.
The titration schedule was identical to the acute trials: xanomeline/trospium 50/20mg BID for the first two days, 100/20mg BID on days 3-7, then flexible dosing up to a maximum of 125/30mg BID starting on day 8, with the option to return to 100/20mg based on tolerability.
Outcomes
Before digging into the numbers we have to address the dropout rate. Of the 566 individuals who were part of the safety population (i.e. got at least 1 dose), only 277 (49%) completed the 52-week study period, and 17.7% left the study due to adverse effects. It will be important to remember that when we discuss the data at 52-weeks, we are only talking about the data from the 277 individuals that completed the entire study.
Should we be concerned about this 49% dropout rate and the 17% discontinuation rate because of treatment-emergent adverse events (TEAEs)? Does it suggest that Xanomeline is a uniquely bad drug and that patients don’t really like it?
Eh, not really.
Remember, this study used patients who were stable on another antidopaminergic prior to entry, but there was no cross-taper to Xanomeline. My suspicion is that this would predispose patients to drop-out more frequently than, say, an open-label extension of a double-blind RCT.
This is supported by the fact that 30% of all dropouts occurred by Week 4 post-switch, and 47% of all dropouts occurred within the first 8 weeks of this 52 week study.
I think this early period reflects a confluence of a few things:
(1) I think the Cobenfy titration schedule is probably just too aggressive for a large chunk of patients; ~18% reported vomiting! I remember being told by a Bristol Myers Squibb drug rep shortly after Xanomeline got its marketing approval not to use this rapid up-titration schedule because it was not well tolerated and instead to allow for a week or so on the lowest dose.
(2) Cobenfy’s onset of action seems like it is roughly equivalent to the antidopaminergics (i.e. you need 4-6 weeks to get to full effect). Some of these dropouts are probably due to the fact that, due to the lack of cross-titration, their psychotic symptoms got worse and they (understandably!) weren’t willing to roll the dice to see if things would improve. This is supported by the fact that “schizophrenia” + “psychotic disorder” adverse event codes accounted for 3.2% of all dropouts. For reference, “nausea” was 5.1% and “vomiting” was 4.1%.
This suggests to me that there is potentially room to improve patient retention in the real-world by (1) doing a slower titration and (2) either cross-titrating or not doing a washout. These seem like fixable problems!
Second, when we look at other open-label studies of antidopaminergics with similar lengths, we see very similar dropout rates. This 52-week study of paliperidone had a 53% dropout rate, with 7% for TEAEs. This 22-month lurasidone extension study had a 65% dropout rate at 12-months. Basically, there’s nothing here to suggest that Xanomeline is particularly poorly tolerated.
Nothing New, And That’s Great
Primary outcomes were safety measures: treatment-emergent adverse events (TEAEs), vital signs, and laboratory parameters. Each TEAE was assigned a severity2 and a likelihood of being related to treatment.3
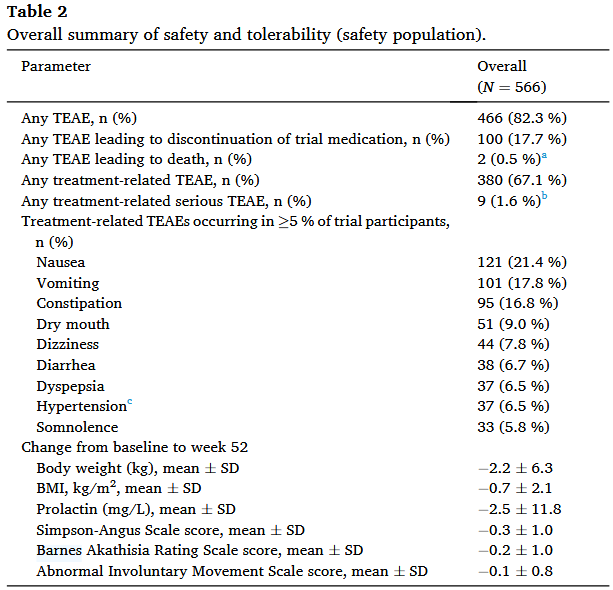
Let’s take a peek at these outcomes in Table 2:
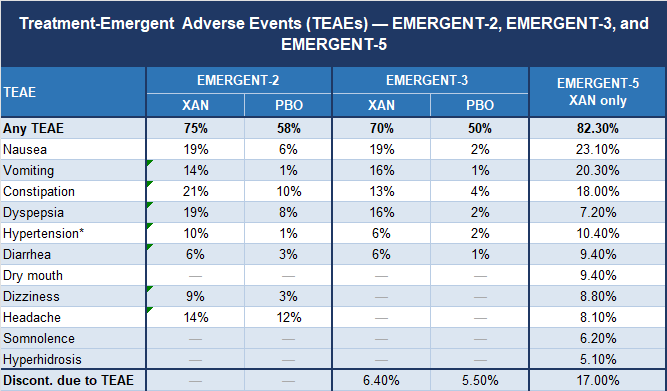
And now that same table but next to the same data from EMERGENT-2 and EMERGENT-3:
I would chalk up the significantly different discontinuation rates to the fact that EMERGENT-2/3 were conducted in a population of inpatients who were not being switched off of an antipsychotic that was already keeping them somewhat stable.
Otherwise, there’s nothing new here. This is a good thing.
It’s “Hypertension” not Hypertension
There’s some nuance with the “hypertension” TEAE that is worth going into. Basically, TEAE tracking uses a coding system called MedDRA to classify them into categories. This mostly works, but MedDRA doesn’t capture continuous variables like blood pressure very well, so the MedDRA definition of “hypertension” encompasses both a transient bump of +5mmHg systolic and a sustained increase of +20mmHg systolic. I think the data suggests that this is “hypertension” and not hypertension:
Mean change from baseline in supine systolic BP peaked at week 1 (+2.7 mmHg) and was +1.8 mmHg at week 52… For supine HR, mean change from baseline peaked at week 2 (+7.6 beats per minute) and was +2.4 beats per minute at week 52. Mean systolic BP, diastolic BP, and HR showed similar patterns between supine and standing measurements…
37/566 (6.5 %) participants had a treatment-related hypertension TEAE. The majority of these hypertension TEAEs were mild or moderate in severity (36/37), transient (29/37), and did not lead to trial discontinuation (37/37).
One patient with pre-existing hypertension did experience tachycardia and a nonserious AV block, so don’t write off the potential for some more serious cardiovascular side-effects.
Still No EPS or Metabolic Side-Effects
It continues to be extremely heartening to see that there are really no EPS to speak of in these patients. 1 year should be long enough to catch everything except for tardive dyskinesia. Considering that we think that TD is secondary to prolonged D2 blockade and none of the other EPS that are also associated with D2 blockade are anywhere to be seen, I would bet large sums of money that TD will just never show up with Xanomeline monotherapy.
Similarly, there continues to be no sign of metabolic side-effects. The average participant lost about 2.2kg (σ: 6.3), which means about 30% lost >5kg. There was no sign of meaningful change in HDL, LDL, triglycerides, or HbA1c. Again, all good news.
I am a little confused about this though. Shouldn’t we see more changes than just an improvement in weight, considering we’re stopping antidopaminergics, which are known to cause metabolic derangements? Maybe these changes are stickier than I realized.
Efficacy
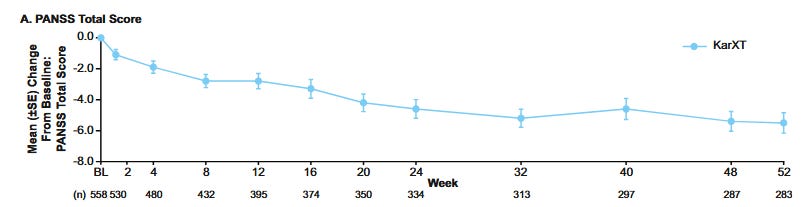
I’m really not going to spend much time on the efficacy data here, because I’m not sure there’s terribly much to be gleaned from an open-label study without a placebo control.
You might be tempted to draw positive conclusions when looking at this graph but remember (1) the baseline phase was after they discontinued their medications in an (up-to) 14-day screening phase, so their PANSS probably rose a bit; (2) the patients who stuck with the drug were probably the ones who saw greater benefit relative to their previous antidopaminergic.
I think the best we can say is that, there’s no clear sign that switching to Xanomeline makes people catastrophically worse, but that’s not really much of a surprise given the RCT data.
Still Looks Good To Me
Frankly, this paper doesn’t contain anything all that interesting, and that’s probably a good thing for Xanomeline. My tl;dr:
About 50% of patients who were stable on an antidopaminergic did not tolerate the switch to Xanomeline
Tolerability could probably be improved by:
Cross-tapering or immediately switching drugs instead of having a washout period.
More slowly increasing the dose
Side-effect profile is the same as it was in the RCTs
Gastrointestinal side-effects predominate
Clinically meaningful cardiovascular side-effects are extremely unusual (0.1%)
No EPS, weight gain, or metabolic side-effects
Nothing to suggest that the efficacy story in the RCTs is not true in the general population
It is perhaps worth wondering what these sorts of patients look like! Presumably many of them have something they dislike about their current medication.
Mild, moderate, or severe
Unrelated, unlikely, possibly, or definitely related to Xanomeline treatment


I agree that the 50% dropout rate is misleading relative to our typical clinical situation. I’ve also found the titration schedule on the label to be too fast for tolerability. I also suspect that real world practice includes a lot more cross-tapering and off-label augmentation which wasn’t captured by the 52-week OLE. I have been getting some akathisia, but this is confounded by polypharmacy
It will be interesting to see, as things move forward, how much the GI side effects become a barrier to its use. While the availability of Cobenfy may be useful for patients with pre-existing obesity, diabetes, high triglycerides, etc., it would be problematic if it causes vomiting resulting in patients vomiting up all their meds, or the constipation/diahorrea results in medicine toxicity/vitamin deficiencies etc.
Side effects like nausea, constipation, diahorrea, can be a real barrier to taking a medication. Such symptoms make daily life difficult and can prevent a patient from even being able to leave the house. So, Cobenfy may help a proportion of patients towards some form of stability but a subset of those same patients may be in the miserable position of having to weigh that against never being 5 secs away from a loo, or unable to be in a car due to nausea.